|
“Heat can not itself flow from colder body to a hotter body.” “It is impossible to construct a device (operating in a cycle) that can transfer heat from cold body to the hot body without absorbing any work.” “It is impossible to construct a device (operating in a cycle) which works on a single heat source and converts all of its heat completely into work” “In all the spontaneous processes, the entropy of the universe increases.” So let me share with you some glimpse of the statement of second law of thermodynamics here only.Įntropy statement of Second law of thermodynamics: I know you will not visit this above article. If you do not know anything about the second law of thermodynamics then I suggest you to refer this detailed article on the second law of thermodynamics which will make you understand all the three statements of the 2nd law. Well, let’s get started with some amazing real life examples of second law of thermodynamics. I’ll just show you the images like this and you are done with all the examples.  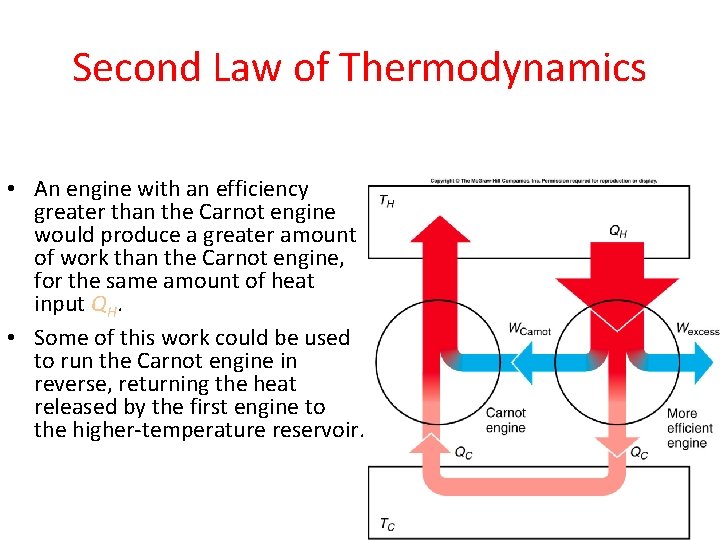
All the examples of second law of thermodynamics are very easy. While it can not become hot again on its own.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
